Writing a CAPA report is one of the most critical skills for quality professionals in regulated industries. Whether you're in pharmaceuticals, medical devices, aerospace, or manufacturing, understanding how to create effective corrective and preventive action plans can make the difference between regulatory compliance and costly violations. Studies show that inadequate CAPA procedures are consistently the leading cause of FDA 483 observations, with CAPA deficiencies representing approximately 12-15% of all FDA inspection citations and appearing in roughly 58% of medical device warning letters. This comprehensive guide will walk you through every step of writing your first CAPA report with confidence.
What is a CAPA Report and Why Does It Matter?
A corrective and preventive action report is a formal document that identifies quality issues, analyzes their root causes, and establishes systematic actions to eliminate problems and prevent their recurrence. More than just paperwork, CAPA reports serve as the backbone of continuous improvement in quality management systems, directly impacting product safety, regulatory compliance, and operational efficiency.
The CAPA process addresses two distinct but interconnected actions: corrective actions that eliminate existing nonconformities and preventive actions that prevent potential issues from occurring. This dual approach ensures organizations not only fix current problems but also strengthen their systems against future failures.
Understanding Corrective Actions vs Preventive Actions
Corrective actions are reactive measures taken to eliminate the causes of existing nonconformities or undesirable situations. These actions focus on addressing problems that have already occurred, such as product defects, process failures, or customer complaints. The goal is to prevent recurrence by attacking the root cause rather than just treating symptoms.
Preventive actions, conversely, are proactive measures designed to eliminate potential nonconformities before they manifest. These actions identify and address weaknesses in processes, systems, or procedures that could lead to problems. Preventive actions often stem from trend analysis, risk assessments, or lessons learned from other organizations or products.
When Your Organization Needs a CAPA Report
Organizations should initiate CAPA requirements when systematic issues emerge that could impact product quality, safety, or regulatory compliance. Key triggers include customer complaints showing patterns, internal audit findings, regulatory inspection observations, product recalls or field failures, and supplier quality issues affecting multiple batches.
Not every deviation or nonconformance requires a full CAPA investigation. The decision should be based on risk assessment, considering factors like patient safety impact, regulatory significance, and potential for recurrence. Organizations should establish clear CAPA procedures that define when to escalate issues beyond simple corrections to formal CAPA investigations.
Essential Elements of Every CAPA Report Format
A well-structured CAPA report format ensures comprehensive documentation while meeting regulatory requirements. Every effective CAPA report contains standardized sections that guide investigators through systematic problem-solving while creating an audit trail that demonstrates due diligence to regulators and stakeholders.
Required Documentation Components
The corrective action report format must include several critical elements to meet regulatory standards. The problem identification section clearly defines what went wrong, when it occurred, where it was discovered, and its potential impact. This section should provide sufficient detail for any reader to understand the issue without requiring additional context.
The investigation section documents the methodology used to analyze the problem, including data sources, personnel interviewed, and analytical tools employed. Root cause analysis results must demonstrate a systematic approach to identifying underlying causes rather than surface-level symptoms. The corrective and preventive actions section outlines specific, measurable steps with assigned responsibilities and target completion dates.
Regulatory Compliance Standards
CAPA quality requirements vary by industry and region, but several common standards apply across regulated sectors. FDA 21 CFR Part 820.100 mandates that medical device manufacturers establish procedures for implementing corrective and preventive action. The regulation requires analyzing quality data sources, investigating nonconformity causes, identifying necessary actions, verifying effectiveness, and implementing procedural changes.
ISO 13485 for medical devices and ISO 9001 for quality management systems also establish CAPA system requirements. These standards emphasize the importance of data analysis, trend identification, and systematic approaches to problem resolution. Companies must demonstrate that their CAPA processes integrate with other quality system elements and contribute to continuous improvement objectives.
Streamline Your CAPA Process Today – Try BPR Hub’s Compliance Platform
📍 Book a Demo
📧 hello@bprhub.com
The 8-Step CAPA Process for Beginners
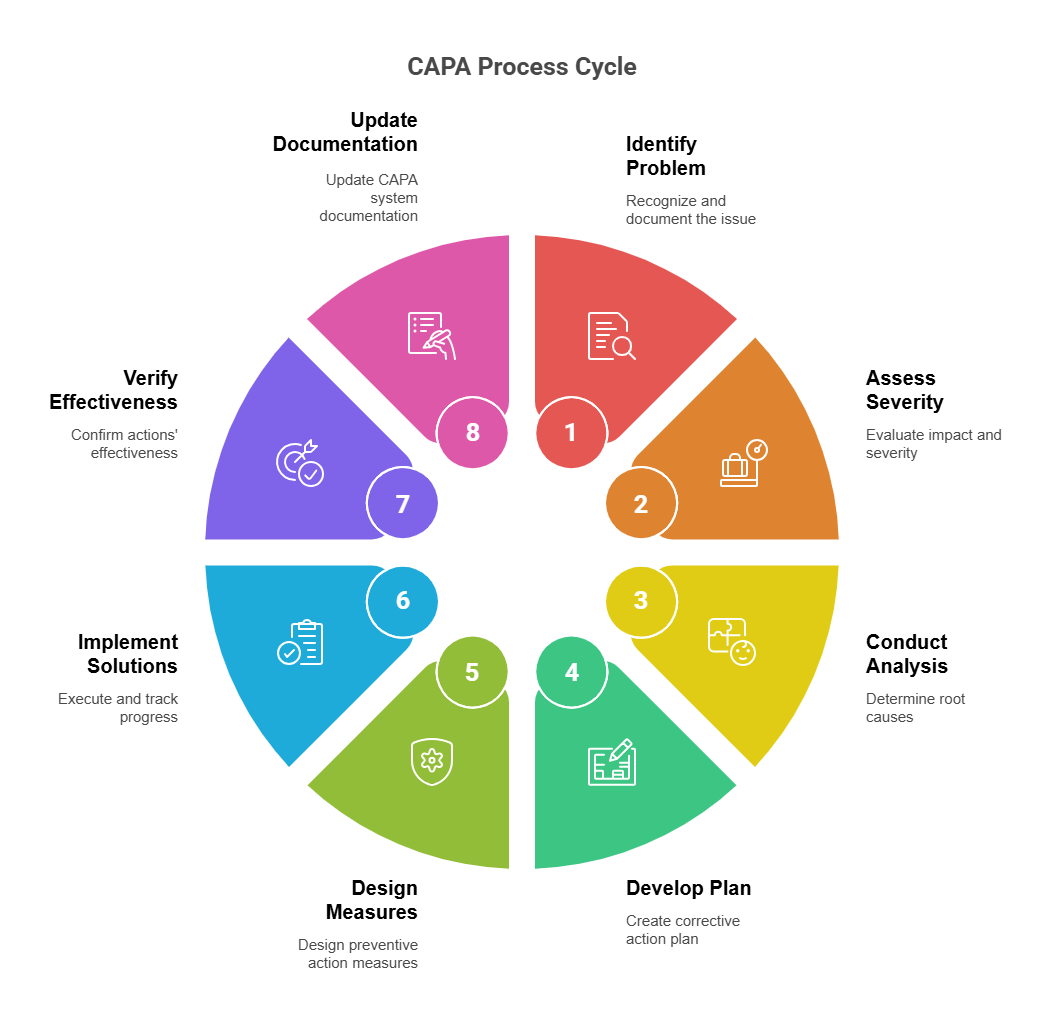
Implementing an effective CAPA process requires a structured approach that ensures thorough investigation and sustainable solutions. This eight-step methodology provides a comprehensive framework for addressing quality issues systematically while meeting regulatory expectations and driving continuous improvement.
Step 1: Identify and Document the Problem
Problem identification forms the foundation of effective CAPA management. Begin by clearly describing what occurred, including specific details about the deviation, nonconformity, or quality issue. Document when the problem was discovered, where it occurred, and any immediate containment actions taken to prevent further impact.
Gather all relevant evidence, including batch records, test results, customer complaints, or inspection reports. Create a clear problem statement that defines the scope and boundaries of the investigation. This statement should be specific enough to focus the investigation while broad enough to capture all related issues that may share common root causes.
Step 2: Assess Issue Severity and Impact
Evaluate the significance of the identified problem using risk-based criteria that consider patient safety, regulatory impact, and business consequences. Establish priority levels that determine resource allocation and timeline expectations for resolution. High-risk issues involving patient safety or regulatory compliance require immediate attention and accelerated timelines.
Consider both immediate and potential long-term impacts of the problem. Assess whether similar issues have occurred previously and evaluate the effectiveness of any past corrective actions. This assessment helps determine the appropriate level of investigation rigor and the scope of corrective actions needed.
Step 3: Conduct Root Cause Analysis
Root cause analysis represents the most critical phase of the CAPA process. Use systematic methodologies such as the 5 Whys, Ishikawa (fishbone) diagrams, or fault tree analysis to identify underlying causes rather than symptoms. Avoid jumping to conclusions or accepting surface-level explanations that may not address systemic issues.
Assemble a cross-functional investigation team with relevant expertise and direct knowledge of the affected processes. Gather data from multiple sources, including process records, personnel interviews, and system documentation. Challenge assumptions and dig deeper until you identify root causes that, if eliminated, would prevent recurrence of the problem.
Step 4: Develop Your Corrective Action Plan
Create a corrective action plan that directly addresses identified root causes with specific, measurable actions. Each action should have clear success criteria, assigned responsibilities, and realistic timelines for completion. Ensure corrective actions are proportionate to the risk and impact of the problem while being sustainable within your organization's resources.
Consider both immediate fixes and long-term solutions. Immediate actions may include process changes, additional controls, or enhanced monitoring. Long-term solutions might involve system redesign, infrastructure improvements, or organizational changes. Document the rationale behind each proposed action and how it addresses specific root causes.
Step 5: Design Preventive Action Measures
Preventive action planning extends beyond the immediate problem to prevent similar issues from occurring in other areas or under different conditions. Analyze your organization's processes, products, and systems to identify where similar root causes might exist. Develop actions that strengthen overall system resilience rather than just fixing the specific problem.
Consider process standardization, improved training programs, enhanced monitoring systems, or design changes that eliminate opportunities for human error. Preventive actions should create lasting improvements that reduce the likelihood of similar problems occurring anywhere within your organization's operations.
Step 6: Implement Solutions and Track Progress
Execute corrective and preventive actions according to established timelines while monitoring progress against defined milestones. Maintain clear communication with all responsible parties and stakeholders throughout implementation. Document any challenges encountered and adjustments made to the original plans.
Establish interim checkpoints to verify that actions are being implemented as planned and are producing expected results. Create feedback loops that allow for course corrections if initial approaches prove ineffective. Track both completion status and early indicators of effectiveness throughout the implementation phase.
Step 7: Verify Effectiveness of Actions
Verification of effectiveness ensures that implemented actions actually solve the problem and prevent recurrence. Establish specific criteria and methods for measuring success, including timeframes for evaluation. This might include statistical analysis of quality data, trend monitoring, or performance indicators that demonstrate improvement.
Design verification activities that provide objective evidence of effectiveness. This could include follow-up inspections, data analysis comparing before and after implementation, or customer feedback indicating problem resolution. Document verification results clearly and address any gaps or shortcomings identified during evaluation.
Step 8: Update Your CAPA System Documentation
Complete the CAPA lifecycle by updating relevant CAPA system procedures, work instructions, and training materials to reflect lessons learned. Ensure that successful solutions are incorporated into standard operating procedures and that knowledge is preserved for future reference. Share insights with other areas of the organization that might benefit from similar improvements.
Conduct a final review of the entire CAPA process to identify opportunities for improving future investigations. Document best practices and lessons learned that can enhance the overall effectiveness of your organization's CAPA program. Close the CAPA formally only after all actions are complete and effectiveness is verified.
CAPA Report Template and Structure
A standardized corrective action report template ensures consistency across investigations while meeting regulatory documentation requirements. The template should guide users through each required section while providing flexibility to address unique aspects of different types of problems. Effective templates balance comprehensiveness with usability, making it easier for investigators to create thorough reports.
Header Information and Identification Numbers
Begin every CAPA report with essential identification information, including a unique CAPA report number, initiation date, and investigation team details. Include the responsible department, priority level, and target completion date. This header information should immediately identify the scope and urgency of the investigation to any reader.
Reference related documents such as deviation reports, customer complaints, or audit findings that triggered the CAPA. Establish clear links to other quality system records that provide additional context or supporting evidence. Include revision control information to track document updates throughout the investigation lifecycle.
Problem Description Section
Writing Clear Issue Statements
Develop concise yet comprehensive problem statements that define the specific issue without interpretation or speculation. Focus on observable facts and measurable impacts rather than opinions or assumptions. The problem statement should enable any reader to understand what occurred without requiring additional context or institutional knowledge.
Include relevant details such as product lots affected, process steps involved, and timeframes when the problem occurred. Specify the discovery method and any immediate containment actions taken. Avoid technical jargon that might obscure understanding for reviewers from different departments or backgrounds.
Including Supporting Evidence
Attach or reference all relevant supporting documentation, including batch records, test results, customer communications, and photographic evidence. Organize evidence logically and ensure it directly supports the problem description and subsequent analysis. Label all attachments clearly and explain their relevance to the investigation.
Maintain original documents in their native format when possible while providing summaries or extracts that highlight key information. Ensure evidence is authentic, complete, and maintains a chain of custody for regulatory review. Consider both direct evidence of the problem and circumstantial evidence that provides context.
Root Cause Analysis Documentation
Document the root cause analysis methodology used and justify its selection based on the problem type and complexity. The present analysis results in a logical flow that shows how you progressed from symptoms to underlying causes. Use visual aids such as fishbone diagrams or fault trees when they enhance understanding.
Provide evidence supporting each potential cause considered and explain why certain possibilities were ruled out. Show how the investigation team validated root causes through data analysis, testing, or other verification methods. Address any disagreements or uncertainties encountered during the analysis process.

Corrective Action Details
Immediate Actions Taken
Document any immediate containment or corrective actions implemented to prevent further impact while the investigation proceeded. Explain the rationale for these actions and their effectiveness in controlling the situation. Include timelines showing when actions were taken and by whom.
Describe how immediate actions relate to long-term solutions and whether temporary measures will be replaced by permanent fixes. Provide evidence that immediate actions were effective and didn't create unintended consequences elsewhere in the system.
Long-term Solutions Planned
Detailed systematic changes are designed to eliminate root causes permanently. Each long-term solution should clearly address specific root causes identified during analysis. Include implementation timelines, resource requirements, and success criteria for each proposed solution.
Explain how long-term solutions will be monitored and maintained over time. Address any risks or challenges associated with implementation and describe contingency plans if primary solutions prove ineffective. Connect long-term solutions to broader organizational improvement objectives.
Preventive Measures Section
Outline actions designed to prevent similar problems from occurring in other areas or under different conditions. Preventive measures should demonstrate learning that extends beyond the immediate problem to strengthen overall system resilience. Show how preventive actions complement corrective actions to create comprehensive risk mitigation.
Address potential applications of lessons learned to other products, processes, or locations within the organization. Include plans for sharing insights with relevant stakeholders and updating procedures or training programs to prevent similar issues elsewhere.
Timeline and Responsibility Assignments
Create clear timelines with specific milestones and deliverables for all corrective and preventive actions. Assign responsibility for each action to specific individuals or roles, ensuring accountability throughout implementation. Include both start dates and completion targets with a realistic buffer time for potential delays.
Establish review points and escalation procedures for actions that fall behind schedule. Define reporting requirements and communication protocols to keep stakeholders informed of progress. Build in flexibility for adjustments while maintaining overall project momentum toward resolution.
Real CAPA Report Example Breakdown
Learning from actual CAPA report examples provides valuable insights into effective documentation practices and common challenges faced during investigations. These examples illustrate how theoretical principles apply to real-world situations while highlighting critical success factors for different types of quality issues.
Common CAPA Procedure Mistakes to Avoid
Understanding typical CAPA procedure pitfalls helps organizations develop more effective processes while avoiding regulatory scrutiny. These mistakes often result from rushed investigations, inadequate training, or failure to allocate sufficient resources to the CAPA process. Learning to recognize and prevent these common errors significantly improves CAPA effectiveness.
Inadequate Root Cause Investigation
The most frequent mistake in CAPA procedures involves superficial root cause analysis that addresses symptoms rather than underlying causes. Organizations often rush to solutions without thoroughly understanding why problems occurred, leading to ineffective corrective actions and problem recurrence. This approach wastes resources and creates false confidence that issues are resolved.
Teams frequently stop investigating once they identify an immediate cause, failing to ask deeper questions about systemic failures. For example, attributing a labeling error solely to "human error" without examining process design, training adequacy, or environmental factors that contributed to the mistake. Effective investigations continue until they identify root causes within management's control to fix.
Establish investigation standards that require multiple analytical techniques and cross-functional team input. Mandate evidence-based conclusions supported by data analysis rather than opinions or assumptions. Create review checkpoints where senior personnel evaluate investigation depth before proceeding to solution development.
Vague Corrective Action Descriptions
Many CAPA systems suffer from poorly defined corrective actions that lack specificity, measurable outcomes, or clear implementation guidance. Actions described as "improve training" or "enhance procedures" provide no meaningful direction for implementation teams and make effectiveness verification impossible. Such vague descriptions indicate inadequate planning and often lead to ineffective solutions.
Effective corrective actions follow SMART criteria (Specific, Measurable, Achievable, Relevant, Time-bound) with clear success criteria and verification methods. Each action should answer what will be done, who will do it, when it will be completed, and how success will be measured. Include sufficient detail that implementation teams can proceed without requiring additional interpretation or clarification.
Missing Preventive Action Components
Organizations often focus exclusively on corrective actions while neglecting preventive action opportunities that could prevent similar problems elsewhere. This narrow focus misses chances to improve overall system resilience and may leave similar vulnerabilities unaddressed in other areas. Preventive actions demonstrate organizational learning and contribute to continuous improvement objectives.
Develop preventive actions that address broader applications of lessons learned beyond the immediate problem scope. Consider how root causes might exist in other products, processes, or locations within the organization. Create systematic approaches for sharing insights and implementing improvements across similar operations.
Poor Documentation Practices
Inadequate documentation represents a critical failure that undermines the entire CAPA process. Poor documentation includes incomplete records, missing evidence, unclear writing, and insufficient detail for regulatory review. Such documentation failures create compliance risks and make it difficult for future investigators to learn from past experiences.
Establish documentation standards that ensure clarity, completeness, and regulatory compliance. Implement review processes that verify documentation quality before CAPA closure. Create templates and training programs that help investigators produce professional, comprehensive reports that meet organizational and regulatory standards.
CAPA Management Best Practices
Effective CAPA management requires systematic approaches that integrate corrective and preventive actions with broader quality management objectives. Best practices focus on creating robust processes, fostering organizational learning, and continuously improving CAPA effectiveness through performance measurement and stakeholder engagement.
Setting Up Effective Review Processes
Establish regular management review processes that evaluate CAPA system performance and identify improvement opportunities. These reviews should examine CAPA metrics, resource allocation, and effectiveness trends while ensuring alignment with organizational quality objectives. Regular review creates accountability and demonstrates management's commitment to quality improvement.
Management reviews should include cross-functional representation to ensure diverse perspectives and comprehensive evaluation. Review agendas should address both individual CAPA status and systemic performance indicators such as closure rates, effectiveness measures, and resource utilization. Use these reviews to identify training needs, process improvements, and resource requirements.
Training Your Team on CAPA Writing
CAPA training programs should develop both technical investigation skills and documentation competencies. Training should cover root cause analysis methodologies, regulatory requirements, and effective writing techniques for creating clear, comprehensive reports. Hands-on exercises using real examples help build practical skills that apply directly to workplace situations.
Develop role-specific training that addresses different responsibilities within the CAPA process. Investigation team members need skills in analytical techniques and evidence evaluation, while reviewers and approvers need skills in quality assessment and regulatory compliance verification. Regular refresher training ensures skills remain current with evolving requirements and best practices.
Integrating with Quality Management Systems
Successful CAPA systems integrate seamlessly with other quality management system elements, including document control, training management, and change control processes. This integration ensures that CAPA actions result in permanent improvements rather than temporary fixes that gradually decay over time.
Link CAPA outcomes to procedure updates, training program modifications, and process improvements that become part of standard operations. Ensure that lessons learned from CAPA investigations inform risk assessments, process validation activities, and continuous improvement programs. Create feedback loops that help the organization learn from both successes and failures in CAPA implementation.
Tools and Resources for CAPA Success
Modern CAPA software solutions and analytical tools significantly enhance investigation efficiency and documentation quality. These tools automate routine tasks, standardize processes, and provide analytical capabilities that improve root cause identification and effectiveness verification. Selecting appropriate tools should balance functionality with user adoption and organizational needs.
Software Solutions for CAPA Tracking
Leading CAPA management platforms offer comprehensive functionality for tracking investigations from initiation through closure. Key features include automated workflow management, document control integration, reporting dashboards, and performance analytics. Solutions like SafetyCulture provide mobile accessibility with free versions for small teams, while enterprise platforms like MasterControl offer extensive integration capabilities.
Cloud-based solutions offer advantages in terms of accessibility, scalability, and automatic updates, while on-premise systems may better serve organizations with specific security or integration requirements. Evaluate solutions based on regulatory compliance features, user interface design, integration capabilities with existing systems, and vendor support quality.
Templates and Checklists
Standardized templates ensure consistency across investigations while reducing the learning curve for new team members. Effective templates provide structure without constraining investigation flexibility, including all required sections, while allowing customization for specific problem types. Templates should incorporate regulatory requirements and organizational standards.
Develop template libraries that address different types of investigations, such as customer complaints, internal audit findings, and supplier quality issues. Include guidance notes and examples within templates to help users understand requirements and expectations. Regular template updates should reflect lessons learned and evolving regulatory requirements.
Regulatory Reference Materials
Maintain current libraries of regulatory guidance documents, industry standards, and best practice resources that support CAPA procedures. Key references include FDA guidance documents, ISO standards, and industry-specific guidelines that define requirements and expectations for corrective and preventive actions.
Organize reference materials by topic and industry sector to facilitate quick access during investigations. Establish procedures for updating reference libraries as regulations change and new guidance becomes available. Ensure investigation teams have access to relevant references and understand how to apply regulatory requirements to specific situations.
Measuring CAPA Effectiveness
CAPA effectiveness measurement provides objective evidence of program success while identifying opportunities for continuous improvement. Key performance indicators should balance efficiency metrics with quality outcomes, measuring both how well the process functions and how effectively it solves problems and prevents recurrence.
Key Performance Indicators
Essential CAPA KPIs include closure rate percentages, average resolution times, and problem recurrence rates. Closure rates measure the percentage of CAPAs completed within target timeframes, typically aiming for 90% or higher on-time completion. Average resolution times help identify bottlenecks and resource constraints, with targets varying by problem complexity and organizational capability.
Problem recurrence rates indicate CAPA effectiveness in addressing root causes, calculated as the percentage of closed CAPAs that don't experience similar problems within a defined follow-up period. Additional metrics might include investigation quality scores, stakeholder satisfaction ratings, and cost-benefit analysis of implemented solutions.
Follow-up and Monitoring Schedules
Establish systematic follow-up procedures that verify corrective action effectiveness over time rather than just at implementation completion. Follow-up schedules should extend sufficiently to capture potential problem recurrence while being practical for resource allocation. Typical follow-up periods range from 3-12 months, depending on problem severity and solution complexity.
Create monitoring systems that track relevant performance indicators and alert management to potential issues before they require new CAPA investigations. Automated monitoring through quality systems can provide early warning of trends that might indicate CAPA effectiveness problems or emerging quality issues.
Continuous Improvement Integration
Link CAPA systems to broader continuous improvement programs that leverage lessons learned for organizational development. Effective integration ensures that CAPA outcomes inform process improvements, training programs, and strategic quality initiatives rather than remaining isolated problem-solving activities.
Regular analysis of CAPA data should identify systematic issues, trending problems, and improvement opportunities that extend beyond individual investigations. Use this analysis to guide resource allocation, training priorities, and process enhancement projects that strengthen overall quality system performance.
How BPR Hub Helps CAPA Report
BPR Hub transforms CAPA management for manufacturing organizations through an integrated compliance platform that streamlines quality processes while ensuring regulatory adherence. The platform automates up to 80% of compliance tasks and reduces certification time by 40%, enabling teams to focus on value-added activities rather than administrative burdens.
BPR Hub's comprehensive approach integrates CAPA procedures with document control, audit management, and training systems, creating seamless workflows that prevent information silos and ensure consistency across quality processes. The platform's AI-driven document analysis and real-time monitoring capabilities help organizations identify potential issues before they require formal CAPA investigations.
The solution supports over 30 manufacturing standards, including ISO 9001, ISO 13485, and AS9100, providing unified compliance management that eliminates the complexity of managing multiple certification requirements separately. This integrated approach particularly benefits organizations managing complex quality systems across multiple locations and product lines.
Eliminate Quality Issues Before They Escalate – Get Started with BPR Hub
📍 Book a Demo
📧 hello@bprhub.com
Key Takeaway
→ CAPA reports serve as the foundation of effective quality management, requiring systematic approaches to problem identification, root cause analysis, and solution implementation
→ Successful corrective and preventive action plans balance immediate problem resolution with long-term system improvements that prevent recurrence and strengthen organizational resilience
→ CAPA procedures must integrate with broader quality management systems to ensure sustainable improvements rather than temporary fixes that gradually decay over time
→ Effective CAPA management relies on clear documentation standards, cross-functional collaboration, and performance measurement to demonstrate regulatory compliance and drive continuous improvement
→ Modern CAPA software solutions and standardized templates significantly enhance investigation efficiency while ensuring consistency and regulatory compliance across all quality investigations
→ Organizations achieve the greatest CAPA effectiveness when they view the process as a strategic improvement tool rather than just a regulatory requirement, leveraging lessons learned to strengthen overall operational excellence
FAQ
Q. How do you write a CAPA report?
Start by clearly identifying and documenting the problem with specific details about what occurred, when, and where. Conduct a thorough root cause analysis using systematic methodologies like 5 Whys or fishbone diagrams. Develop specific corrective actions to eliminate root causes and preventive actions to prevent similar issues elsewhere. Include clear timelines, responsibilities, and verification methods for all actions. Document everything thoroughly with supporting evidence and ensure regulatory compliance throughout.
Q. What should be included in a CAPA plan?
A comprehensive CAPA plan must include a problem description with supporting evidence, root cause analysis results, specific corrective and preventive actions with assigned responsibilities and timelines, implementation procedures, effectiveness verification methods, and documentation requirements. Each section should provide sufficient detail for implementation teams to proceed without additional interpretation while meeting regulatory documentation standards for audit readiness.
Q. What is the difference between corrective and preventive action?
Corrective actions are reactive measures that eliminate the causes of existing problems to prevent recurrence, while preventive actions are proactive measures that address potential issues before they occur. Corrective actions respond to nonconformities that have already happened, whereas preventive actions identify and mitigate risks or weaknesses that could lead to future problems. Both work together to create comprehensive quality improvement.
Q. How long should a CAPA investigation take?
CAPA investigation timelines vary based on problem complexity and risk level, but most should be completed within 30-90 days. Simple issues typically resolve within 30 days, moderate complexity problems require 30-60 days, and complex systemic issues may need 60-90 days. High-risk problems involving safety or regulatory compliance require accelerated timelines with initial responses within 48-72 hours and investigations completing within 30 days maximum.
Q. What are the FDA requirements for CAPA reports?
FDA 21 CFR Part 820.100 requires medical device manufacturers to analyze quality data sources, investigate nonconformity causes, identify necessary corrective and preventive actions, verify action effectiveness, implement procedural changes, communicate information to responsible parties, and submit relevant information for management review. All activities must be thoroughly documented with evidence supporting conclusions and actions taken to address quality problems.
Get insights that help you minimize risks and maximize profits.
Dive deeper into manufacturing compliance with our free resources.
We get it, compliance can get tough.
Here are some additional resources to help.
We get it, compliance can get tough. Here are some additional resources to help.
Get updates in your inbox

.svg)
%20(1).svg)





%20(1).svg)

.avif)

